연구용
Phenothiazine Dopamine Receptor 길항제
제품 번호S4251
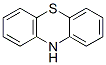
화학 구조
분자량: 199.27
품질 관리
화학 정보, 보관 및 안정성
| 분자량 | 199.27 | 화학식 | C12H9NS |
보관 (수령일로부터) | |
|---|---|---|---|---|---|
| CAS 번호 | 92-84-2 | SDF 다운로드 | 원액 보관 |
|
|
| 동의어 | ENT 38 | Smiles | C1=CC=C2C(=C1)NC3=CC=CC=C3S2 | ||
용해도
|
In vitro |
DMSO
: 40 mg/mL
(200.73 mM)
Water : Insoluble Ethanol : Insoluble |
몰농도 계산기
|
In vivo |
|||||
생체 내 제형 계산기 (투명한 용액)
1단계: 아래 정보 입력 (권장: 실험 중 손실을 고려하여 추가 동물 포함)
2단계: 생체 내 제형 입력 (이것은 계산기일 뿐 제형이 아닙니다. 용해도 섹션에 생체 내 제형이 없는 경우 먼저 당사에 문의하십시오.)
계산 결과:
작업 농도: mg/ml;
DMSO 원액 준비 방법: mg 약물 사전 용해 μL DMSO ( 원액 농도 mg/mL, 농도가 해당 약물 배치의 DMSO 용해도를 초과하는 경우 먼저 당사에 문의하십시오. )
생체 내 제형 준비 방법: 취하다 μL DMSO 원액, 다음 추가μL PEG300, 혼합하고 투명하게 한 다음 추가μL Tween 80, 혼합하고 투명하게 한 다음 추가 μL ddH2O, 혼합하고 투명하게 합니다.
생체 내 제형 준비 방법: 취하다 μL DMSO 원액, 다음 추가 μL 옥수수 기름, 혼합하고 투명하게 합니다.
참고: 1. 다음 용매를 추가하기 전에 액체가 투명한지 확인하십시오.
2. 용매를 순서대로 추가해야 합니다. 다음 용매를 추가하기 전에 이전 추가에서 얻은 용액이 투명한 용액인지 확인해야 합니다. 와동, 초음파 또는 뜨거운 물 중탕과 같은 물리적 방법을 사용하여 용해를 도울 수 있습니다.
작용 메커니즘
| Targets/IC50/Ki |
D2 receptor
|
|---|---|
| 시험관 내(In vitro) |
Phenothiazines는 대부분 10번 위치에 다이알킬아미노알킬 그룹으로, 그리고 2번 위치에 작은 그룹으로 치환되어 신경이완, 항구토, 항히스타민, 항소양, 진통 및 구충과 같은 유익한 활성을 나타냅니다. 2-trifluoromethyl-10-(4-aminobutyl)phenothiazine은 S. cerevisiae 균주와 T. mentagrophites를 각각 0.4 μg/mL 및 1.5 μg/mL의 MIC로 억제합니다. 10-carbamoylalkyl 유도체는 그람 양성균인 Bacillus subtilis에 대해 7.8 μg/mL–30 μg/mL 범위의 MIC로 상당한 활성을 보입니다. 테트라사이클릭 화합물(나프토퀴논 고리로 변형됨)은 S. aureus에 대해 12.5 μg/mL의 MIC50으로 상당한 항균 활성을 보입니다. 부틸렌 링커를 가진 이 화합물들은 프로필렌 링커를 가진 화합물들보다 더 효과적이며, 2-chloro-10-chloroethylureidobutyl 유도체는 4가지 백혈병 세포주와 7가지 대장암 세포주에 대해 1.4 μM 및 1.6 μM의 GI50을 나타냅니다. 10-Amino(hydroxy)propyl 유도체(5 μM)는 2일간의 배양 후 인간 변형 WI38VA 세포에서 세포사멸이 뒤따르는 G2/M기 세포 주기 정지를 유도합니다. 이 종류의 약물은 주로 고리 수산화, 고리 설폭시화, N-탈메틸화, N-산화, 황산염 및 글루쿠론산 접합을 통해 배설되기 전에 체내에서 광범위한 대사를 거칩니다. 이들은 dopamine D2 receptors 및 α1-adrenoceptors보다 α2-adrenoceptors에 대한 결합 친화도가 상당히 낮습니다. 이 화합물들은 M. tuberculosis의 민감성, 다제내성 및 다제내성 균주에 대해 상당한 시험관 내 활성을 가지며, 1차 치료에 사용되는 일부 약제의 활성을 향상시킵니다.
|
참조 |
|
