Mitophagy inhibitors are a class of compounds that specifically block or interfere with the mitophagy process, a selective form of autophagy responsible for the degradation of damaged or dysfunctional mitochondria. As mitophagy plays a crucial role in maintaining cellular homeostasis, regulating energy metabolism, and mediating various pathological processes such as neurodegeneration, cancer, and cardiovascular diseases, the exploration of mitophagy inhibitors has become a frontier topic in biomedical research. These inhibitors not only serve as powerful tools to dissect the molecular mechanisms underlying mitophagy but also hold great potential for the development of novel therapeutic strategies targeting mitophagy-related disorders.
Mitophagy 유도제/활성제 (Mitophagy Inducers/activators)
| Cat.No. | 제품명 | 정보 | 제품 사용 인용 | 제품 검증 |
|---|---|---|---|---|
| S1060 | AZD2281 (Olaparib) | Olaparib (AZD2281, KU0059436)은 무세포 분석에서 5 nM/1 nM의 IC50을 갖는 선택적 PARP1/2 억제제이며, tankyrase-1에 비해 300배 덜 효과적입니다. Olaparib은 BRCA 돌연변이를 가진 세포에서 Mitophagy와 관련된 유의미한 Autophagy를 유도합니다. |
|
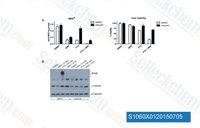
|
| S1378 | Ruxolitinib (INCB18424) | Ruxolitinib (INCB18424)은 세포 없는 분석에서 3.3 nM/2.8 nM의 IC50을 가지며 JAK3에 비해 JAK1/2에 대해 130배 이상의 선택성을 보이는 최초의 강력하고 선택적인 JAK1/2 억제제입니다. 이 화합물은 독성 Mitophagy를 통해 종양 세포를 사멸시킵니다. 이는 Autophagy를 유도하고 Apoptosis related를 향상시킵니다. |
|
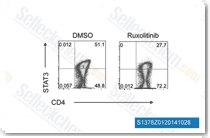
|
| S1208 | Doxorubicin (Adriamycin) Hydrochloride | Doxorubicin (DOX) HCl은 인간 DNA topoisomerase II를 2.67 μM의 IC50으로 억제하는 항생제입니다. Doxorubicin은 AMPK의 기저 인산화를 감소시킵니다. Doxorubicin은 HIV 감염 환자의 동시 치료에 사용되지만 HBV 재활성화 위험이 높은 것으로 나타났습니다.이 제품은 PBS 용액에 용해될 때 침전될 수 있습니다. 순수에 원액을 준비하고 순수 또는 생리 식염수로 희석하여 작업 용액을 얻는 것이 좋습니다.Doxorubicin (Adriamycin) HCl은 신장 질환 동물 모델을 유도하는 데 사용될 수 있습니다. |
|

|
| S1225 | Etoposide | Etoposide는 포도필로톡신의 반합성 유도체로, topoisomerase II 억제 활성을 통해 DNA 합성을 억제하며, 이는 DNA의 이중 가닥 및 단일 가닥 절단을 촉진하고 Topoisomerase II 결합을 통해 복구를 가역적으로 억제합니다. Etoposide는 autophagy, mitophagy 및 apoptosis를 유도합니다. |
|

|
| S1950 | Metformin Hydrochloride | Metformin Hydrochloride (1,1-디메틸비구아니드 염산염)는 주로 간에서 포도당 신생(간에 의한 포도당 생산)을 억제하여 간세포의 고혈당을 감소시키는 매우 효과적인 혈당 강하제입니다. 또한 단핵 세포의 Mitophagy를 촉진하고 JNK/p38 MAPK 경로 및 GADD153 활성화를 통해 폐암 세포의 Apoptosis를 유도합니다. |
|
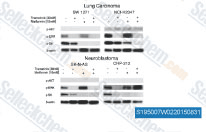
|
| S1792 | Simvastatin (MK-733) | Simvastatin은 무세포 분석에서 0.1-0.2 nM의 Ki를 가진 HMG-CoA Reductase의 경쟁적 억제제입니다. Simvastatin은 ferroptosis, mitophagy, autophagy 및 apoptosis를 유도합니다. |
|
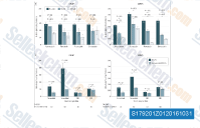
|
| S5243 | Ruxolitinib (INCB18424) Phosphate | Ruxolitinib Phosphate (INCB018424, INC424)는 Ruxolitinib의 인산염 형태입니다. Ruxolitinib은 세포가 없는 조건에서 3.3 nM/2.8 nM의 IC50을 가지며, JAK3에 비해 JAK1/2에 대해 130배 이상의 선택성을 보이는 최초의 강력하고 선택적인 JAK1/2 억제제로 임상에 진입했습니다. Ruxolitinib은 독성 미토파지를 통해 종양 세포를 사멸시킵니다. Ruxolitinib은 Autophagy를 유도하고 Apoptosis를 강화합니다. |
|
|
| S1322 | Dexamethasone | Dexamethasone은 글루코코르티코이드 스테로이드 약물 계열의 강력한 합성 구성원이며, 항염증 및 면역억제 효과를 가지는 인터루킨 수용체 조절자입니다. Dexamethasone은 autophagy 및 mitophagy를 유도합니다. Dexamethasone은 COVID-19로 입원한 환자에게 시험되었으며 중증 환자에게 유익한 것으로 밝혀졌습니다. |
|
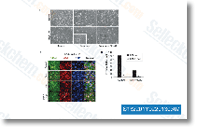
|
| S1759 | Pitavastatin (NK-104) Calcium | 스타틴 계열 약물의 새로운 구성원인 Pitavastatin calcium은 고효율 HMG-CoA 환원효소 억제제인 피타바스타틴의 칼슘염 제제입니다. Pitavastatin Calcium은 ROS 생성 억제를 통해 AGEs 유도 미토파지를 약화시킵니다. Pitavastatin Calcium은 자가포식 및 세포자멸사를 유도합니다. |
|

|
| S2391 | Quercetin (Sophoretin) | 채소, 과일 및 와인에 존재하는 천연 플라보노이드인 Quercetin은 재조합 SIRT1의 자극제이자 IC50 2.4-5.4 μM의 PI3K 억제제입니다. Quercetin은 mitophagy, apoptosis 및 보호 autophagy를 유도합니다. 4단계. |
|
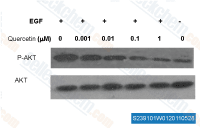
|
Classification of Mitophagy Inhibitors Based on Mitophagy Pathway
Mitophagy is regulated by multiple conserved pathways, and mitophagy inhibitors can be classified into different types according to the specific pathways they target. This classification is essential for understanding the specificity of inhibitors and their applicability in different research scenarios.
Inhibitors Targeting the PINK1-Parkin Pathway
The PINK1-Parkin pathway is the most well-characterized mitophagy pathway, and its dysregulation is closely associated with Parkinson's disease. Inhibitors targeting this pathway mainly act by blocking the activation of PINK1 or Parkin, or interfering with the downstream signaling events. For example, Cyclosporine A (CsA) has been shown to inhibit mitophagy by suppressing the interaction between Parkin and mitochondria, thereby preventing the ubiquitination of mitochondrial proteins that are essential for mitophagy initiation. Another inhibitor, N-acetylcysteine (NAC), exerts its inhibitory effect by scavenging reactive oxygen species (ROS), which are important triggers for PINK1 activation. Research on these inhibitors has greatly contributed to the understanding of the molecular details of the PINK1-Parkin pathway and its role in neurodegenerative diseases.
Inhibitors Targeting Non-PINK1-Parkin Pathways
In addition to the PINK1-Parkin pathway, mitophagy can also be induced through various non-canonical pathways, such as those mediated by BNIP3, NIX, and FUNDC1. Inhibitors targeting these pathways have unique mechanisms of action. For instance, 3-Methyladenine (3-MA), a classic autophagy inhibitor, can also block BNIP3-mediated mitophagy by inhibiting the class III PI3K complex, which is required for the formation of autophagosomes. Similarly, chloroquine (CQ) and hydroxychloroquine (HCQ) inhibit mitophagy by increasing the pH of lysosomes, thereby impairing the fusion between autophagosomes and lysosomes, a process that is essential for the degradation of mitophagosomes regardless of the initiating pathway. These non-specific inhibitors are widely used in research to study the general role of mitophagy in cellular processes, although their lack of pathway specificity needs to be considered when interpreting experimental results.
The Relationship Between Mitophagy Inhibitors and Autophagy Regulation
Mitophagy is a subset of autophagy, and thus, there is a close relationship between mitophagy inhibitors and autophagy regulators. Some mitophagy inhibitors are also general autophagy inhibitors, while others are specific to mitophagy, reflecting the differences in the regulatory mechanisms between general autophagy and selective mitophagy. Understanding this relationship is crucial for selecting appropriate inhibitors in research and avoiding off-target effects.
Overlap Between Mitophagy Inhibitors and General Autophagy Inhibitors
As mentioned earlier, 3-MA, CQ, and HCQ are typical examples of inhibitors that affect both autophagy and mitophagy. These compounds target the core machinery of autophagy, such as autophagosome formation and lysosomal function, which are shared by general autophagy and mitophagy. Therefore, their use in mitophagy research requires careful control experiments to distinguish the specific effects on mitophagy from those on general autophagy. For example, combining these inhibitors with mitophagy-specific markers can help confirm whether the observed effects are indeed due to the inhibition of mitophagy rather than general autophagy.
Specific Mitophagy Inhibitors and Their Independence from General Autophagy
With the deepening of research, an increasing number of mitophagy-specific inhibitors have been identified. These inhibitors selectively block mitophagy without significantly affecting general autophagy, making them ideal tools for studying the specific functions of mitophagy. For example, Mdivi-1, a small molecule inhibitor, specifically targets the mitochondrial fission protein Drp1, which is required for mitophagy but not for general autophagy. By inhibiting Drp1-mediated mitochondrial fission, Mdivi-1 blocks the segregation of damaged mitochondria from the mitochondrial network, thereby preventing their engulfment by autophagosomes. Studies using Mdivi-1 have revealed the specific role of mitophagy in regulating mitochondrial quality control and cellular survival under stress conditions.
Research Methods Involving Mitophagy Inhibitors: Parking and Fasting Models
Parking (Parkin-related) models and fasting models are commonly used in mitophagy research to induce mitophagy, and combining these models with mitophagy inhibitors can provide valuable insights into the physiological and pathological roles of mitophagy. These models simulate different physiological conditions, allowing researchers to study mitophagy in a more relevant biological context.
Mitophagy Inhibitors in Parking Models
Parking models are established by manipulating the expression or activity of Parkin, a key regulator of the PINK1-Parkin mitophagy pathway. These models are widely used to study the role of mitophagy in Parkinson's disease. For example, in Parkin-overexpressing cells or animal models, mitophagy is significantly enhanced. By treating these models with mitophagy inhibitors such as CsA or Mdivi-1, researchers can investigate the consequences of mitophagy inhibition on mitochondrial function, neuronal survival, and disease progression. These studies have provided important evidence for the involvement of mitophagy dysregulation in Parkinson's disease and have laid the foundation for the development of therapeutic strategies targeting mitophagy.
Mitophagy Inhibitors in Fasting Models
Fasting is a physiological stimulus that induces both general autophagy and mitophagy to provide energy for cells during nutrient deprivation. Fasting models are widely used to study the physiological functions of mitophagy. When mitophagy inhibitors are applied in fasting models, they can block the adaptive mitophagy response, leading to the accumulation of damaged mitochondria and impaired energy metabolism. For example, in fasting mice treated with CQ, the levels of mitophagy markers such as LC3-II/LC3-I ratio and p62 degradation are significantly reduced, and the mice show decreased tolerance to fasting due to impaired mitochondrial function. These studies have highlighted the importance of mitophagy in maintaining cellular homeostasis during nutrient stress and have provided insights into the role of mitophagy in metabolic disorders.
Detection of Mitophagy Markers in Research Using Mitophagy Inhibitors
The accurate detection of mitophagy markers is essential for evaluating the efficacy of mitophagy inhibitors and interpreting research results. Mitophagy markers can be divided into two categories: mitochondrial markers and autophagy markers, and their combination is often used to confirm mitophagy inhibition.
Mitochondrial Markers for Mitophagy Detection
Mitochondrial markers such as Tom20, COXIV, and Cytochrome c are commonly used to assess the degradation of mitochondria during mitophagy. When mitophagy is inhibited, the levels of these mitochondrial proteins increase due to the reduced degradation. For example, treatment with Mdivi-1 in cells under mitophagy-inducing conditions leads to the accumulation of Tom20 and COXIV, indicating the inhibition of mitophagy. In addition, mitochondrial DNA (mtDNA) content can also be used as a marker, as mitophagy inhibition results in the accumulation of mtDNA.
Autophagy-Mitophagy Fusion Markers
Autophagy-mitophagy fusion markers, such as the co-localization of LC3 (an autophagosome marker) with mitochondrial markers, are more specific for mitophagy. The use of immunofluorescence or confocal microscopy to detect the co-localization of LC3 and Tom20/COXIV can directly reflect the formation of mitophagosomes. When mitophagy is inhibited by inhibitors such as CQ or Mdivi-1, the co-localization of LC3 and mitochondrial markers is significantly reduced. In addition, the LC3-II/LC3-I ratio and the degradation of p62 (a substrate of autophagy) are also commonly used to assess autophagy and mitophagy activity, although they need to be combined with mitochondrial markers to confirm mitophagy-specific effects.
Interactions Between Mitophagy Inhibitors and Mitophagy Inducers in Research
Mitophagy inducers are compounds that promote mitophagy, and their combination with mitophagy inhibitors is often used in research to study the reversibility of mitophagy and to validate the specificity of inhibitors. This combination also provides a basis for the development of combination therapies targeting mitophagy-related diseases.
Validation of Mitophagy Inhibitor Specificity Using Inducers
Mitophagy inducers such as carbonyl cyanide m-chlorophenyl hydrazone (CCCP) and antimycin A are commonly used to induce mitophagy by causing mitochondrial depolarization. When mitophagy inhibitors are applied together with these inducers, the inhibitory effect of the inhibitors can be evaluated by detecting mitophagy markers. For example, CCCP treatment increases the LC3-II/LC3-I ratio and reduces p62 levels, while co-treatment with Mdivi-1 or CQ reverses these effects, confirming the specificity of the inhibitors. This approach is widely used in the initial screening and validation of new mitophagy inhibitors.
Synergistic and Antagonistic Effects in Therapeutic Research
In therapeutic research targeting mitophagy-related diseases, the combination of mitophagy inhibitors and inducers may exhibit synergistic or antagonistic effects. For example, in cancer research, some cancer cells rely on enhanced mitophagy to survive under hypoxic conditions. The use of mitophagy inhibitors can block this adaptive response, making cancer cells more sensitive to chemotherapy or radiotherapy. On the other hand, mitophagy inducers may be beneficial in neurodegenerative diseases by promoting the clearance of damaged mitochondria. However, the inappropriate use of inhibitors or inducers may have adverse effects, highlighting the need for a comprehensive understanding of the role of mitophagy in specific diseases. The interaction between mitophagy inhibitors and inducers provides important clues for optimizing therapeutic strategies and avoiding potential side effects.
In conclusion, mitophagy inhibitors are essential tools in mitophagy research, providing valuable insights into the molecular mechanisms and physiological functions of mitophagy. The classification based on mitophagy pathways helps in selecting specific inhibitors for different research purposes, while the understanding of their relationship with autophagy regulators ensures the accuracy of research results. The application of mitophagy inhibitors in parking and fasting models simulates physiological and pathological conditions, facilitating the study of mitophagy in relevant biological contexts. The accurate detection of mitophagy markers is crucial for evaluating inhibitor efficacy, and the interaction with mitophagy inducers provides a basis for therapeutic development. Future research should focus on identifying more specific and potent mitophagy inhibitors, exploring their mechanisms of action in depth, and evaluating their therapeutic potential in various mitophagy-related diseases.
