연구용
Pelabresib (CPI-0610) BET Inhibitor
제품 번호S7853
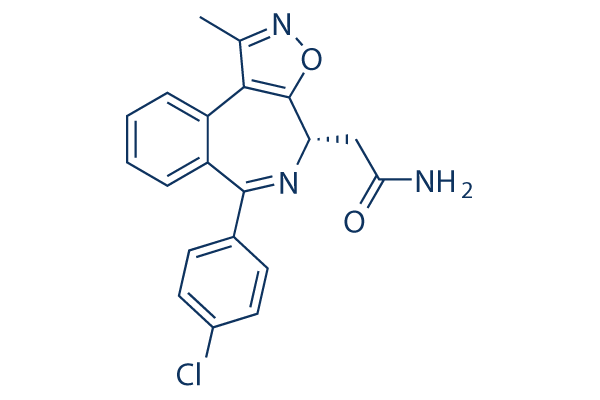
화학 구조
분자량: 365.81
품질 관리
화학 정보, 보관 및 안정성
| 분자량 | 365.81 | 화학식 | C20H16ClN3O2 |
보관 (수령일로부터) | |
|---|---|---|---|---|---|
| CAS 번호 | 1380087-89-7 | SDF 다운로드 | 원액 보관 |
|
|
| 동의어 | N/A | Smiles | CC1=NOC2=C1C3=CC=CC=C3C(=NC2CC(=O)N)C4=CC=C(C=C4)Cl | ||
용해도
|
In vitro |
DMSO
: 73 mg/mL
(199.55 mM)
Ethanol : 12 mg/mL Water : Insoluble |
몰농도 계산기
|
In vivo |
|||||
생체 내 제형 계산기 (투명한 용액)
1단계: 아래 정보 입력 (권장: 실험 중 손실을 고려하여 추가 동물 포함)
2단계: 생체 내 제형 입력 (이것은 계산기일 뿐 제형이 아닙니다. 용해도 섹션에 생체 내 제형이 없는 경우 먼저 당사에 문의하십시오.)
계산 결과:
작업 농도: mg/ml;
DMSO 원액 준비 방법: mg 약물 사전 용해 μL DMSO ( 원액 농도 mg/mL, 농도가 해당 약물 배치의 DMSO 용해도를 초과하는 경우 먼저 당사에 문의하십시오. )
생체 내 제형 준비 방법: 취하다 μL DMSO 원액, 다음 추가μL PEG300, 혼합하고 투명하게 한 다음 추가μL Tween 80, 혼합하고 투명하게 한 다음 추가 μL ddH2O, 혼합하고 투명하게 합니다.
생체 내 제형 준비 방법: 취하다 μL DMSO 원액, 다음 추가 μL 옥수수 기름, 혼합하고 투명하게 합니다.
참고: 1. 다음 용매를 추가하기 전에 액체가 투명한지 확인하십시오.
2. 용매를 순서대로 추가해야 합니다. 다음 용매를 추가하기 전에 이전 추가에서 얻은 용액이 투명한 용액인지 확인해야 합니다. 와동, 초음파 또는 뜨거운 물 중탕과 같은 물리적 방법을 사용하여 용해를 도울 수 있습니다.
작용 메커니즘
| Targets/IC50/Ki |
BRD4-BD1
(Cell-free assay) 39 nM
MYC
(in MV-4-11 cells) 180 nM(EC50)
|
|---|---|
| 시험관 내(In vitro) |
Pelabresib (CPI-0610)은 사이토카인 존재 하에서 및 골수 기질 세포와 공동 배양 시 MM(다발성 골수종) 세포 성장을 억제합니다. 이는 MYC 하향 조절과 관련된 세포자멸사 및 G1 세포 주기 정지를 유도합니다. 그러나 BET 억제 시 MM 세포에서 BCL2, NF-κB 및 MCL1의 단백질 수준은 변하지 않습니다. 이 화합물은 MM 세포에서 전사와 단백질 수준 모두에서 Ikaros 및 IRF4 발현을 억제합니다. |
| 생체 내(In vivo) |
MV-4-11 급성 골수성 백혈병 세포를 사용한 마우스 이종이식 모델에서 Pelabresib (CPI-0610) 처리 마우스의 MYC mRNA 수준은 4시간째에 대조군에 비해 상당히 감소했으며, 이후 시점에서는 혈장 내 이 화합물의 자유 농도 감소와 일치하여 대조군 수준으로 회복되었습니다. BET bromodomain 억제는 검사 기간 동안 종양 성장을 상당히 억제했습니다(각각 41%, 80%, 74%의 종양 성장 억제). 동물에게서 유의미한 체중 감소는 없었습니다. 쥐와 개에서 허용 가능한 독성 프로파일을 기반으로, 두 종 모두에서 공통적인 독성이 관찰되었습니다: 림프계 고갈; 골수 저형성증과 관련된 빈혈 및 혈소판 감소증; 위장관 점막 위축, 미란 및 궤양; 고환 정세관 상피의 퇴행; 경증에서 중등도의 고혈당증. 이러한 독성은 가역적입니다. |
참조 |
|
임상시험 정보
(데이터 출처 https://clinicaltrials.gov, 업데이트 날짜 2024-05-22)
| NCT 번호 | 모집 | 조건 | 스폰서/협력자 | 시작일 | 단계 |
|---|---|---|---|---|---|
| NCT05391022 | Completed | Advanced Malignancies|Solid Tumor|Hematological Malignancy |
Constellation Pharmaceuticals |
July 20 2021 | Phase 1 |
| NCT02986919 | Withdrawn | Peripheral Nerve Tumors |
University of Texas Southwestern Medical Center |
May 5 2017 | Phase 2 |
| NCT02157636 | Completed | Multiple Myeloma |
Constellation Pharmaceuticals|The Leukemia and Lymphoma Society |
July 15 2014 | Phase 1 |
| NCT01949883 | Completed | Lymphoma |
Constellation Pharmaceuticals|The Leukemia and Lymphoma Society |
September 5 2013 | Phase 1 |
