연구용
Pinometostat (EPZ5676) DOT1L 억제제
제품 번호S7062
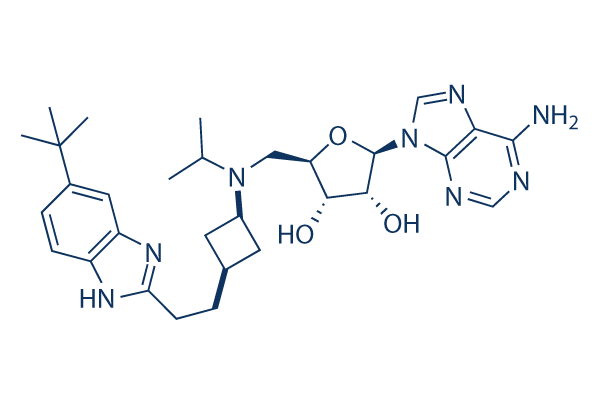
화학 구조
분자량: 562.71
품질 관리
함께 자주 사용되는 제품 Pinometostat (EPZ5676)
| 관련 타겟 | HDAC JAK BET PKC PARP HIF PRMT EZH2 AMPK Histone Acetyltransferase |
|---|---|
| 기타 Histone Methyltransferase 억제제 | 3-Deazaneplanocin A (DZNep) Hydrochloride BIX-01294 trihydrochloride UNC1999 EPZ015666 (GSK3235025) EPZ004777 MM-102 (HMTase Inhibitor IX) Chaetocin SGC 0946 EPZ005687 UNC0638 |
세포 배양, 처리 및 작업 농도
| 세포주 | 분석 유형 | 농도 | 배양 시간 | 제형 | 활성 설명 | PMID |
|---|---|---|---|---|---|---|
| MV4-11 | Function assay | 4 days | Inhibition of DOT1L in human MV4-11 cells expressing MLL-AF4 assessed as reduction of H3K79me2 level after 4 days by ELISA method | 25406853 | ||
| MOLM13 | Proliferation assay | Antiproliferative activity against human MOLM13 cells containing MLL-AF9, EC50=4 nM | 23879463 | |||
| MV4-11 | Proliferation assay | Antiproliferative activity against human MV4-11 cells containing MLL-AF4, EC50=4 nM | 23879463 | |||
| THP1 | Proliferation assay | Antiproliferative activity against human THP1 cells containing MLL-AF9, EC50=4 nM | 23879463 | |||
| HeLa | Function assay | 72 hrs | Inhibition of DOT1L in human HeLa cells assessed as reduction in H3K79me2 level after 72 hrs by ELISA, IC50 = 0.007 μM. | 28337327 | ||
| MV4-11 | Antiproliferative assay | 6 hrs | Antiproliferative activity against human MV4-11 cells harboring MLL-AF4 treated for 6 hrs measured after 8 days by Celltiter-Glo reagent based assay, IC50 = 0.015 μM. | 28337327 | ||
| MOLM13 | Function assay | 72 hrs | Inhibition of DOT1L in human MOLM13 cells assessed as suppression of HoxA9 gene after 72 hrs by luciferase reporter gene assay, IC50 = 0.052 μM. | 28337327 | ||
| 클릭하여 더 많은 세포주 실험 데이터 보기 | ||||||
화학 정보, 보관 및 안정성
| 분자량 | 562.71 | 화학식 | C30H42N8O3 |
보관 (수령일로부터) | |
|---|---|---|---|---|---|
| CAS 번호 | 1380288-87-8 | SDF 다운로드 | 원액 보관 |
|
|
| 동의어 | N/A | Smiles | CC(C)N(CC1C(C(C(O1)N2C=NC3=C(N=CN=C32)N)O)O)C4CC(C4)CCC5=NC6=C(N5)C=C(C=C6)C(C)(C)C | ||
용해도
|
In vitro |
DMSO
: 100 mg/mL
(177.71 mM)
Ethanol : 25 mg/mL Water : Insoluble |
몰농도 계산기
|
In vivo |
|||||
생체 내 제형 계산기 (투명한 용액)
1단계: 아래 정보 입력 (권장: 실험 중 손실을 고려하여 추가 동물 포함)
2단계: 생체 내 제형 입력 (이것은 계산기일 뿐 제형이 아닙니다. 용해도 섹션에 생체 내 제형이 없는 경우 먼저 당사에 문의하십시오.)
계산 결과:
작업 농도: mg/ml;
DMSO 원액 준비 방법: mg 약물 사전 용해 μL DMSO ( 원액 농도 mg/mL, 농도가 해당 약물 배치의 DMSO 용해도를 초과하는 경우 먼저 당사에 문의하십시오. )
생체 내 제형 준비 방법: 취하다 μL DMSO 원액, 다음 추가μL PEG300, 혼합하고 투명하게 한 다음 추가μL Tween 80, 혼합하고 투명하게 한 다음 추가 μL ddH2O, 혼합하고 투명하게 합니다.
생체 내 제형 준비 방법: 취하다 μL DMSO 원액, 다음 추가 μL 옥수수 기름, 혼합하고 투명하게 합니다.
참고: 1. 다음 용매를 추가하기 전에 액체가 투명한지 확인하십시오.
2. 용매를 순서대로 추가해야 합니다. 다음 용매를 추가하기 전에 이전 추가에서 얻은 용액이 투명한 용액인지 확인해야 합니다. 와동, 초음파 또는 뜨거운 물 중탕과 같은 물리적 방법을 사용하여 용해를 도울 수 있습니다.
작용 메커니즘
| Targets/IC50/Ki |
DOT1L
(Cell-free assay) 80 pM(Ki)
|
|---|---|
| 시험관 내(In vitro) |
EPZ-5676은 MV4-11 세포에서 2.6 nM의 세포 IC50으로 H3K79 디메틸화를 감소시킵니다. EPZ-5676 치료는 다른 히스톤 부위의 메틸화 상태에 영향을 미치지 않으면서 H3K79 메틸화의 농도 및 시간 의존적 감소를 유도하며, 이는 핵심 MLL 표적 유전자의 억제와 MLL 재배열 백혈병 세포에서 선택적 세포자멸사적 세포 사멸로 이어집니다. EPZ-5676은 MLL-AF4 재배열 세포주 MV4-11의 증식을 9 nM의 IC50으로 억제합니다. |
| 생체 내(In vivo) |
MLL 재배열 백혈병 이종이식 모델에 21일 동안 EPZ-5676을 지속적으로 정맥 주입한 결과 용량 의존적인 항종양 활성을 보였습니다. 최고 용량인 70.5 mg/kg/일에서는 치료 중단 후 최대 32일 동안 재성장 없이 완전한 종양 퇴행이 달성되었습니다. 효능 연구 동안 EPZ-5676로 치료한 쥐에서는 유의미한 체중 감소나 명백한 독성이 관찰되지 않았습니다. |
참조 |
|
적용 분야
| 방법 | 바이오마커 | 이미지 | PMID |
|---|---|---|---|
| Western blot | p-p65 / p65 / NFATc1 CDK6 / BCL11A / Bcl-2 / RUNX1 / MEF2C / H3K79me3 / H4 |
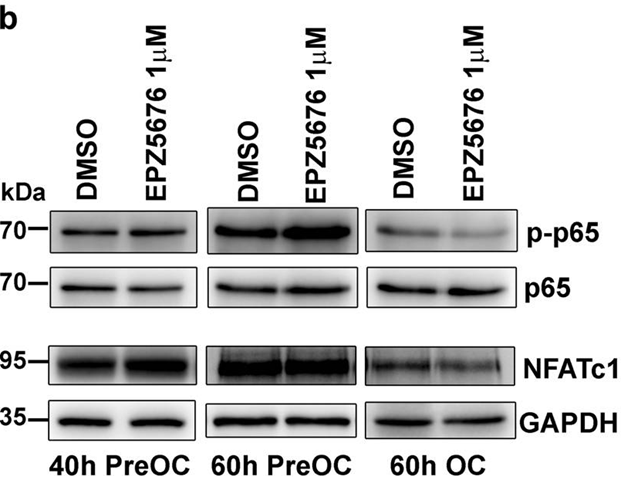
|
29348610 |
| Immunofluorescence | NFATc1 |
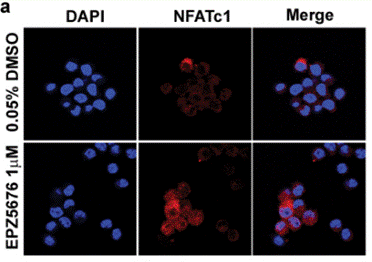
|
29348610 |
임상시험 정보
(데이터 출처 https://clinicaltrials.gov, 업데이트 날짜 2024-05-22)
| NCT 번호 | 모집 | 조건 | 스폰서/협력자 | 시작일 | 단계 |
|---|---|---|---|---|---|
| NCT02141828 | Completed | Leukemia|Acute Myeloid Leukemia|Acute Lymphocytic Leukemia|Acute Leukemias |
Epizyme Inc.|Celgene Corporation|Ipsen |
May 2014 | Phase 1 |
| NCT01684150 | Completed | Acute Myeloid Leukemia|Acute Lymphoblastic Leukemia|Myelodysplastic Syndrome|Myeloproliferative Disorders |
Epizyme Inc.|Celgene|Ipsen |
September 2012 | Phase 1 |
기술 지원
자주 묻는 질문
질문 1:
Is the vehicle 30% PEG400/0.5% Tween80/5% propylene glycol recommended for in vivo use?
답변:
S7062 in 30% PEG400/0.5% Tween80/5% propylene glycol at 30 mg/ml is a suspension. For injection, 2% DMSO/30% PEG 300/5% Tween 80/ddH2O at 5 mg/ml is suitable.
